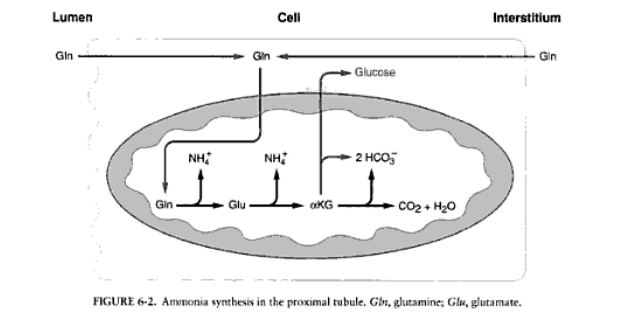
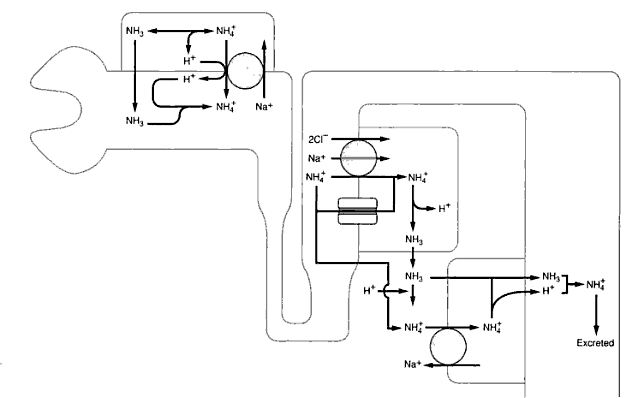
Role of Ammonia Buffer in Urinary System
1. Role in production stages by also producing new HCO3- and glucose
2. Role in acidification by combining with H+
Alkali Reserve
Limiting pH
The limiting pH is the minimum pH that is achievable by the body acidification system. The maximum gradient by which the nephrons can secrete H+ ion is 1000 times the H+ concentration in plasma. Since the pH of plasma is about 7.5, the H+ concentration is
(H+) of plasma=10^(-7.5)
since we know, pH=-log(H+)
The maximum gradient of urine H+ is therefore= 1000*10^(-7.5)=10^(-4.5)
Therefore, the pH of urine is=pH 4.5
This is the limiting pH
(H+) of plasma=10^(-7.5)
since we know, pH=-log(H+)
The maximum gradient of urine H+ is therefore= 1000*10^(-7.5)=10^(-4.5)
Therefore, the pH of urine is=pH 4.5
This is the limiting pH